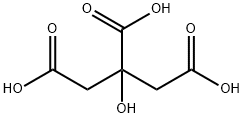
Citric acidCAS#77-92-9
Citric acid is a white, crystalline, weak organic acid present in most plants and many animals as an intermediate in cellular respiration. Citric acid contains three carboxyl groups making it a carboxylic, more specifically a tricarboxylic, acid.the name citrus originates from the Greek kedromelon meaning apple of melon for the fruit citron. Greek works mention kitron, kitrion, or kitreos for citron fruit, which is an oblong fruit several inches long from the scrublike tree Citrus medica. Lemons and limes have high citric acid content, which may account for up to 8% of the fruit's dry weight.
| Citric acid Chemical Properties |
| Melting point | 153-159 °C (lit.) |
| Boiling point | 310 °C (decomp) |
| bulk density | 560kg/m3 |
| density | 1.67 g/cm3 at 20 °C |
| vapor density | 7.26 (vs air) |
| vapor pressure | <0.1 hPa (20 °C) |
| refractive index | 1.493~1.509 |
| FEMA | 2306 | CITRIC ACID |
| Fp | 100 °C |
| storage temp. | 2-8°C |
| solubility | Citric acid also dissolves in absolute (anhydrous) ethanol (76 parts of citric acid per 100 parts of ethanol) at 15 °C. |
| form | grit |
| pka | 3.14(at 20℃) |
| color | White |
| Odor | Odorless |
| PH | 3.24(1 mM solution);2.62(10 mM solution);2.08(100 mM solution); |
| Odor Type | odorless |
| biological source | synthetic |
| explosive limit | 8%, 65°F |
| Water Solubility | soluble in Water (1174g/L at 10°C, 1809g/L at 30°C, 3825g/L at 80°C). |
| Sensitive | Hygroscopic |
| λmax | λ: 260 nm Amax: 0.20 λ: 280 nm Amax: 0.10 |
| Merck | 14,2326 |
| JECFA Number | 218 |
| BRN | 782061 |
| Henry's Law Constant | 3.1×1015 mol/(m3Pa) at 25℃, Burkholder et al. (2019) |
| Stability: | Stable. Incompatible with bases, strong oxidizing agents, reducing agents, metal nitrates. |
| Cosmetics Ingredients Functions | CHELATING FRAGRANCE BUFFERING |
| Cosmetic Ingredient Review (CIR) | Citric acid (77-92-9) |
| InChI | 1S/C6H8O7/c7-3(8)1-6(13,5(11)12)2-4(9)10/h13H,1-2H2,(H,7,8)(H,9,10)(H,11,12) |
| InChIKey | KRKNYBCHXYNGOX-UHFFFAOYSA-N |
| SMILES | OC(=O)CC(O)(CC(O)=O)C(O)=O |
| LogP | -1.64 |
| CAS DataBase Reference | 77-92-9(CAS DataBase Reference) |
| NIST Chemistry Reference | 1,2,3-Propanetricarboxylic acid, 2-hydroxy-(77-92-9) |
| EPA Substance Registry System | Citric acid (77-92-9) |
| Safety Information |
| Hazard Codes | Xi,C,T |
| Risk Statements | 41-36/37/38-36/38-37/38-34-36-35-61-60 |
| Safety Statements | 26-39-37/39-24/25-36/37/39-45-36-53 |
| RIDADR | UN 1789 8/PG 3 |
| WGK Germany | 1 |
| RTECS | GE7350000 |
| F | 9 |
| TSCA | TSCA listed |
| HS Code | 2918 14 00 |
| Storage Class | 11 - Combustible Solids |
| Hazard Classifications | Eye Irrit. 2 STOT SE 3 |
| Hazardous Substances Data | 77-92-9(Hazardous Substances Data) |
| Toxicity | LD50 in mice, rats (mmol/kg): 5.0, 4.6 i.p. (Gruber, Halbeisen) |
Product Application of CAS#77-92-9
Citric acid is a weak organic acid that is known as a commodity chemical, as more than a million tonnes are produced every year by mycological fermentation on an industrial scale using crude sugar sol utions, such as molasses and strains of Aspergillus niger. Citric acid is widely distributed in plants and in animal tissues and fluids and exist in greater than grace amounts in variety of fruits and vegetables, most notably in citrus fruits such as lemon and limes. Citric acid is mainly used as an acidifier, flavoring agent and chelating agent. It was also used as a chemical restrainer particularly in developers for the collodion process and in silver nitrate solutions used for sensitizing salted and albumen papers.
Fast delivery time
Inventory 2-3 working days New production 7-10 working days