Dextrose CAS#50-99-7
CAS: 50-99-7
MF: C6H12O6
MW: 180.16
EINECS: 200-075-1
MOQ (Minimum Order Quantity): 1 FCL (Full Container Load)
Dextrose CAS#50-99-7
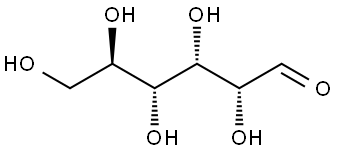
glucose ,a short form of dextrorotatory glucose, is a stereoisomer of glucose molecule, which is biologically active and whose bottom chiral carbon has its hydroxyl group (OH) located spatially to the right. Its molecule can exist in an open-chain (acyclic) and ring (cyclic) form and has two isomers α- and β-. It is the main source of energy in the form of ATP for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. In animals, it arises from the breakdown of glycogen in a process known as glycogenolysis. D-(+)-Glucose has been used as a standard for the estimation of total sugar in hydrolyzed starch by phenol-sulfuric acid method. It has also been used in the preparation of the liquid media for culturing some yeast cells. In addition, it is used therapeutically in fluid and nutrient replacement, such as glucose syrup and glucose powder. It can be obtained by enzymatic cleavage of starch, so there are multiple sources like sugar cane, sugar beet, corn (corn syrup), potatoes and wheat. Today, large-scale starch hydrolysis is used to produce glucose.
| Melting point | 150-152 °C(lit.) |
| alpha | 52.75 º (c=10, H2O, NH4OH 25 ºC) |
| Boiling point | 232.96°C (rough estimate) |
| density | 1.5440 |
| bulk density | 630kg/m3 |
| refractive index | 53 ° (C=10, H2O) |
| storage temp. | room temp |
| solubility | H2O: 1 M at 20 °C, clear, colorless |
| pka | pKa 12.43(H2O,t = 18,)(Approximate) |
| form | Crystalline Powder |
| color | White |
| PH | 5.0-7.0 (25℃, 1M in H2O) |
| Odor | Odorless |
| PH Range | 5.9 |
| biological source | wheat |
| Optical Rotation | [α]25/D +52.5 to +53.0°(lit.) |
| Water Solubility | Soluble |
| λmax | λ: 260 nm Amax: 0.03 λ: 280 nm Amax: 0.02 |
| Merck | 14,4459 |
| BRN | 1281608 |
| Henry's Law Constant | 7.8×108 mol/(m3Pa) at 25℃, Qin et al. (2021) |
| Stability: | Stable. Substances to be avoided include strong oxidizing agents. Combustible. |
| Cosmetics Ingredients Functions | HUMECTANT |
| InChI | 1S/C6H12O6/c7-1-2-3(8)4(9)5(10)6(11)12-2/h2-11H,1H2/t2-,3-,4+,5-,6+/m1/s1 |
| InChIKey | WQZGKKKJIJFFOK-DVKNGEFBSA-N |
| SMILES | OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@@H]1O |
| LogP | -2.490 (est) |
| CAS DataBase Reference | 50-99-7(CAS DataBase Reference) |
| NIST Chemistry Reference | Glucose(50-99-7) |
| EPA Substance Registry System | Dextrose (50-99-7) |
| Safety Information |
| Hazard Codes | Xi,Xn |
| Risk Statements | 36/37/38-63-62-46-36/38-21 |
| Safety Statements | 26-36/37-24/25-53-25 |
| WGK Germany | 1 |
| RTECS | LZ6600000 |
| F | 3 |
| Autoignition Temperature | 500 °C |
| TSCA | TSCA listed |
| HS Code | 17023051 |
| Storage Class | 11 - Combustible Solids |
| Hazardous Substances Data | 50-99-7(Hazardous Substances Data) |
| Toxicity | LD50 orally in Rabbit: 25800 mg/kg |
Product Application of Dextrose CAS#50-99-7
Glucose is the primary fuel for biological respiration. During digestion, complex sugarsand starches are broken down into glucose (as well as fructose and galactose) in the small intestine.Glucose then moves into the bloodstream and is transported to the liver where glucoseis metabolized through a series of biochemical reactions, collectively referred to as glycolysis.Glycolysis, the breakdown of glucose, occurs in most organisms. In glycolysis, the final productis pyruvate. The fate of pyruvate depends on the type of organism and cellular conditions.In animals, pyruvate is oxidized under aerobic conditions producing carbon dioxide. Underanaerobic conditions in animals, lactate is produced. This occurs in the muscle of humansand other animals. During strenuous conditions the accumulation of lactate causes musclefatigue and soreness. Certain microorganisms, such as yeast, under anaerobic conditions convertpyruvate to carbonic dioxide and ethanol. This is the basis of the production of alcohol.Glycolysis also results in the production of various intermediates used in the synthesis of otherbiomolecules. Depending on the organism, glycolysis takes various forms, with numerousproducts and intermediates possible.
Fast delivery time
Inventory 2-3 working days New production 7-10 working days